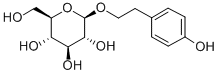
RHODIOLOSIDE; RHODIOLOSIDE.Beta - d - glucopyranoside, 2 - (4 - hydroxyphenyl) baton rouge.Glucopyranoside, p -
hydroxyphenethyl;Rhodosin;2 - (4 - HYDROXYPHENYL) baton rouge - BETA - D - GLUCOPYRANOSIDE;TWEEN 20 EXTRA PURE;SALIDROSIDE 98+% BY HPLC;RhodiolaCrenulataExtract;SALIDROSIDE (P);Rhodiola Rosea (1% 3% Rosavin, Salidroside);Rhodiola Extract;Rhodiola rosea extract;2 - (4 - hydroxyphenyl) baton rouge beta - D - glucopyranoside
|
Product Name
|
Specification
|
Test Method
|
|
Rhodiola Rosea Extract
|
Salidroside≥3%
|
By HPLC
|
|
Rhodiola Rosea Extract
|
Salidroside≥5%
|
By HPLC
|
|
Rhodiola Rosea Extract
|
Salidroside≥10%
|
By HPLC
|
|
Rhodiola Rosea Extract
|
Salidroside≥98%
|
By HPLC
|
|
Rhodiola Rosea Extract
|
Rosavins(Rosavin+ Rosarin+ Rosin)≥5%
Rosavin≥1%
|
By HPLC
|
|
Rhodiola Rosea Extract
|
Rosavins(Rosavin+ Rosarin+ Rosin)≥5%
Rosavin≥2%
|
By HPLC
|
|
Rhodiola Rosea Extract
|
Rosavins(Rosavin+ Rosarin+ Rosin)≥3%
Salidroside≥1%
|
By HPLC
|
|
Rhodiola Rosea Extract
|
Rosavins(Rosavin+ Rosarin+ Rosin)≥5%
Salidroside≥2%
|
By HPLC
|
|
ANALYSIS
|
SPECIFICATION
|
METHOD
|
|
Assay
|
Salidroside≥3.0%
|
HPLC
|
|
Organoleptic
|
|
Appearance
|
Red brown powder
|
Visual
|
|
Odor
|
Characteristic
|
Visual
|
|
Tasted
|
Characteristic
|
Organoleptic
|
|
Physical Characteristics
|
|
Sieve Analysis
|
95% pass 80 mesh
|
EP7.0
|
|
Loss on Drying
|
≤5.0%
|
EP7.0
|
|
Ash
|
≤5.0%
|
EP7.0
|
|
Solvent Residues
|
|
Methanol
|
≤1000ppm
|
USP35
|
|
Ethanol
|
≤25ppm
|
USP35
|
|
Heavy Metals
|
|
Total Heavy Metals
|
≤10ppm
|
Atomic Absorption
|
|
As
|
≤2ppm
|
Atomic Absorption
|
|
Pb
|
≤3ppm
|
Atomic Absorption
|
|
Cd
|
≤1ppm
|
Atomic Absorption
|
|
Hg
|
≤0.1ppm
|
Atomic Absorption
|
|
Microbiology
|
|
Total Plate Count
|
≤1000CFU/g
|
USP35
|
|
Yeast & Mold
|
≤100CFU/g
|
USP35
|
|
E.Coli
|
Negative/g
|
USP35
|
|
Salmonella
|
Negative/g
|
USP35
|
Appliction
1. As Food and beverage ingredients.
2. As Healthy Products ingredients.
3. As Nutrition Supplements ingredients.
4.As a health food and cosmetic ingredients.